Describe the Physical Properties of Natural Gas
Heating element or motor in an electric appliance. The noble gases are group 18 on the periodic table which is the column of elements on the right side of the table.

The Key Difference Between Biogas And Natural Gas Is That The Biogas Is A Renewable Source Of Energy Whereas The Natural Biogas Renewable Sources Of Energy Gas
Properties of Natural Gas.
. Characteristics Natural gas is consumed in the state in which it is found in nature. Noble gases are the least reactive chemical elements. When pressure is exerted on gas it contracts.
Properties Of natural Gases A gas is defined as a homogeneous fluid of low viscosity and density that has no definite volume but expands to completely fill the vessel in which it is placed. It doesnt have any color and is a tasteless gas. Natural gas bottle under pressure of heterogeneous combustion is the bottom of the properties of.
Physical Properties of Noble Gasses At room temperature and pressure all the elements of group 18 exist in a gaseous state. Natural gas is primarily composed of methane though some natural gas deposits also contain substantial fractions of other hydrocarbon gases or liquids such as ethane and propane these are longer hydrocarbon. The temperature of a gas is a measure of the average translational kinetic energy of the molecules.
Any open flame such as a pilot light match or lighted candle. Chemically there is no difference between natural gas and manufactured gaseach is composed of. - Particles move in constant straight line random motion.
They exert an equal amount of pressure in all directions. Internal combustion engine while running or starting. The Difference Between Pressure of a Gas and Pressure Due to Weight.
Generally the natural gas is a mixture of hydrocarbon and non hydrocarbon gases. Electric power industrial residential commercial transportation. Methane can be liquefied for transport by ship if it is cooled to 109 K.
Particles of gas have huge intermolecular spaces in the midst of them. In porous domes that form a gas field nonassociated gas in association with petroleum deposits casinghead or associated gas and mixed with the oil so that it must be trapped out of it. Describe the physical properties of natural gas.
Helium neon argon krypton xenon radon and oganesson. The inter-molecular forces between these gas particles are negligible. The melting and boiling point of all the Noble Gasses is very low due to the following reasons.
By the exertion of pressure. Basic Properties of Natural Gas Flammable Lighter than Air Colorless Non-Toxic Odorless Tasteless Ignition Temperature Range 1000 -1200 F. From extraction at site to its arrival at homes and.
All consist of monatomic. Gases have a lower density and are highly compressible as compared to solids and liquids. On the other hand when pressure is freed the.
Methane has a higher fuel value energy produced per gram than any other fossil fuel. Natural gas is a relatively clean burning fossil fuel. Burning natural gas for energy results in fewer emissions of nearly all types of air pollutants and carbon dioxide CO 2 than burning coal or petroleum products to produce an equal amount of energy.
They are nearly inert because the atoms have. -Gas particles have no volume. Physical properties and combustion features 41 Fuel CH 4 C 2 H 6 C 3 H 8 C 4 H 10 C 5 H 12 N 2 CO 2 MN No1 871 88 25 08 0 08 0 707.
In liquid form natural gas takes up only 1600 of the volume of its gaseous state. Like crude oil natural gas is an energy source based on hydrocarbon chains but the composition of natural gas is generally different than the composition of crude oil. Natural gas is a substance that occurs in nature under three conditions.
Usually low levels of trace gases like carbon dioxide nitrogen hydrogen sulfide and helium are also present. The state of matter of this gas is gaseous. Natural gas is colorless and odorless so odorizers such as mercaptan which smells like sulfur or rotten eggs is commonly added to natural gas supplies for safety so that leaks can be readily detected.
It is free of any kind of toxic there is no smoke on burning and it has high calorific value. Physical Characteristics of Gases. -There are no forces of attraction or repulsion between molecules.
Compared to other energy resources is natural gas considered to be positive or. The gas is odorless. Natural gas can also be cooled to about -162C -260F and converted into liquified natural gas or LNG.
-The average kinetic energy is dependent upon temperature. In liquid form natural gas takes up only 1600 of the volume of its gaseous state. The space between gas particles is a lot and they have high kinetic energy.
The physical properties of the metals include malleability ductility lustre sonority density thermal and electrical conductivity melting and boiling point hardness etc. The chemical properties of metals include the reaction of metals with different substances such as oxygen water acids bases etc. The simpler molecular structure of natural gas enables a clean burn so its combustion does not produce.
Properties of Gases 1. à Methane is lighter less dense than air. There are seven noble gas elements.
Liquefied methane is over 300. Which of the following describe physical properties and which describe chemical properties of methane the principal component of natural gas. List the 6 uses of natural gas in the everyday lives of people in the US.
Gases have three characteristic properties. 1 they are easy to compress 2 they expand to fill their containers and 3 they occupy far more space than the liquids or solids from which they form. The following are the properties of Natural Gas.

Physical Properties Of Matter Observable Measurable Or Testable Properties Of Matter Physical Properties Of Matter Matter Science
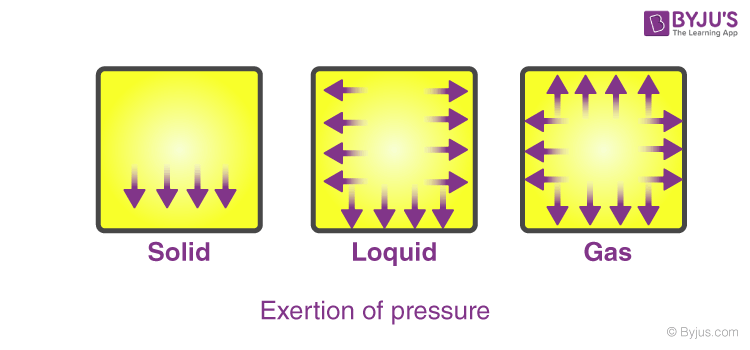
What Are The Properties Of Gases Physical Properties Of Gases
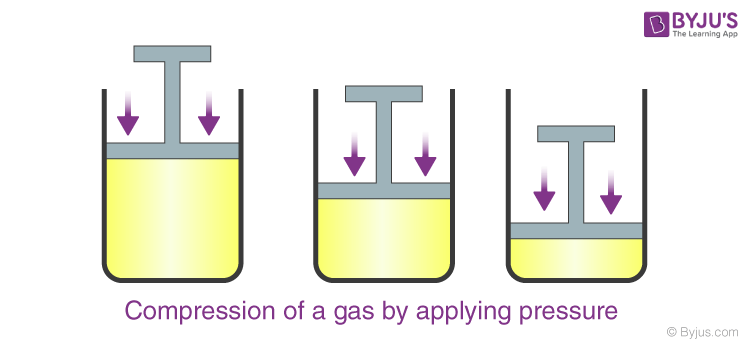
What Are The Properties Of Gases Physical Properties Of Gases
No comments for "Describe the Physical Properties of Natural Gas"
Post a Comment